Edmonton, Alberta, Canada, December 13, 2021 -- Pacylex today announced that data from the first ever clinical trial of an N-myristoyltransferase (NMT) inhibitor was presented at the American Society of Hematology (ASH) Annual Meeting and Exposition from 11-14 December 2021. This update included the design of the first clinical trial with PCLX-001 in Non-Hodgkin’s Lymphoma and solid tumor patients and an update on the first dose cohort. Details of the poster are below.
In addition to the presentation, the abstract was published online in the November supplemental issue of Blood.
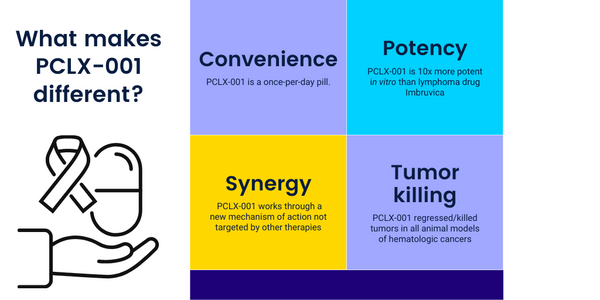
Three patients have been accrued to Dose Level 1 of 20 mg orally once-per-day. No dose-limiting toxicities have been identified. Preliminary pharmacokinetic analysis shows rapid oral absorption, an elimination half-life of ~ 12 hours, and no plasma accumulation. The preliminary PK is consistent with once-per-day oral administration.
“We are very pleased that the first clinical experience with an NMT inhibitor shows the initial dose to be well tolerated and well absorbed, with a PK consistent with a daily oral drug,” said John Mackey, CMO of Pacylex.
All three patients were enrolled at the University of Alberta Cross Cancer Institute. Dr. Randeep Sangha, the principal investigator for the study of PCLX-001 at the Cross Cancer Institute in Edmonton, said “our first experience with PCLX-001 on refractory diffuse large B-cell lymphoma (DLBCL) and Leiomyosarcoma patients was very encouraging and consistent with further exploration of its potential as a new treatment for patients”.
Patient enrollment and dosing will also begin soon at Princess Margaret Hospital in Toronto and the BC Cancer Agency in Vancouver. The study will enroll 20-30 patients in the initial phase. Three principal investigators will oversee the clinical study at the three clinical sites in Canada: Dr. John Kuruvilla at Princess Margaret Cancer Centre in Toronto, Dr. Randeep Sangha at the Cross Cancer Institute in Edmonton, and Dr. Laurie Sehn at the British Columbia Cancer Center in Vancouver.
This study is registered at ClinicalTrials.gov Identifier: NCT04836195.
PCLX-001 (aka DDD86481) is a first-in-class, small molecule NMT inhibitor originally developed by the University of Dundee Drug Discovery Unit as part of a program to treat African sleeping sickness, funded by Wellcome Trust. Pacylex is developing PCLX-001 in the form of a once-a-day pill initially to treat leukemia and lymphoma. PCLX-001 has also been shown to inhibit the growth of lung and breast cancer tumors in animal models. In leukemia, lymphoma, and breast cancer patients, the levels of NMT2 is correlated with survival, suggesting an important biological role in these cancers. In tests using cultured cancer cells in vitro, PCLX-001 is at least ten times as potent as ibrutinib (Imbruvica) and dasatinib (Sprycel), two clinically approved drugs currently used to treat hematologic malignancies.
About Pacylex
Pacylex is a pharmaceutical company targeting hematologic and solid cancers with a new first-in-class therapeutic, headquartered in Edmonton, Alberta, Canada. Pacylex’s technology combines new insights from Dr. Luc Berthiaume of the University of Alberta connecting myristoylation to cancer with a family of high quality myristoylation inhibitors Pacylex licensed from the University of Dundee. PCLX-001 is the lead drug in a new class of NMT inhibitors, enabling Pacylex to exploit NMTs as new clinical targets for cancer treatment. Pacylex initiated clinical studies in Canada in the fall of 2021 in diffuse large B-cell lymphoma and solid tumors. Pacylex is also receiving support from an Alberta Innovates AICE grant in 2020, and the research leading to this breakthrough was supported in part by the Alberta Cancer Foundation and the Cure Cancer Foundation.
For more information:
Pacylex Pharmaceuticals Contact: Michael J. Weickert, CEO of Pacylex Pharmaceuticals, Inc.
E: michael.weickert@pacylex.com
P: 650-218-1840